What You Must Know Before Studying Isomers?
Organic chemistry starts with carbon behaviour. Carbon always forms 4 bonds. That single rule explains most structures you see later. Hydrocarbons are compounds made only of carbon and hydrogen. Among them, alkanes are the simplest group. Key points you should already understand:
- Carbon, carbon single bonds define alkanes
- Each carbon completes four bonds
- Hydrogen fills any remaining bonding space
- No double or triple bonds exist in alkanes
Image:
This bonding pattern gives alkanes a predictable formula.
General Formula of Alkanes
For Any Alkane: CnH₂n+₂
Using this formula helps you check whether a structure is valid.
Example:
4 carbons → C₄H₁₀
That formula belongs to butane
Why Structure Matters More Than Formula?
Many students assume one formula means one compound. That idea breaks here. Carbon atoms can link in more than one way while keeping the same molecular formula. When that happens, different compounds exist. These compounds are called structural isomers of butane because their bonding arrangement changes, not their formula.
Molecular Formula vs. Structural Formula
A molecular formula shows only the number and types of atoms (e.g., ), which can represent different molecules like glucose and fructose that behave differently because their atoms are arranged differently.
Structural Formula Importance
A structural formula shows how atoms are bonded and arranged in space, which determines a molecule’s physical properties, chemical reactivity, and biological function.
Isomers Concept
One molecular formula does not always correspond to one compound because carbon atoms can bond in multiple ways, producing different compounds with identical formulas called isomers.
Butane Example: Butane () exists as two structural isomers with different bonding arrangements, resulting in different boiling points, reactivity, and physical behaviour.
Function Depends on Structure
A molecule’s function is directly determined by its structure, and even small changes in bonding or spatial orientation can significantly affect how it interacts with other substances.
Understanding Isomerism Without Memorisation
An isomer is not created by bending or rotating bonds. Rotation does not change identity. An isomer appears only when:
- The carbon backbone changes
- A branch form
- The connectivity shifts
Ask Yourself! Can I draw the carbon chain differently without breaking bonds? If yes, you may have an isomer.
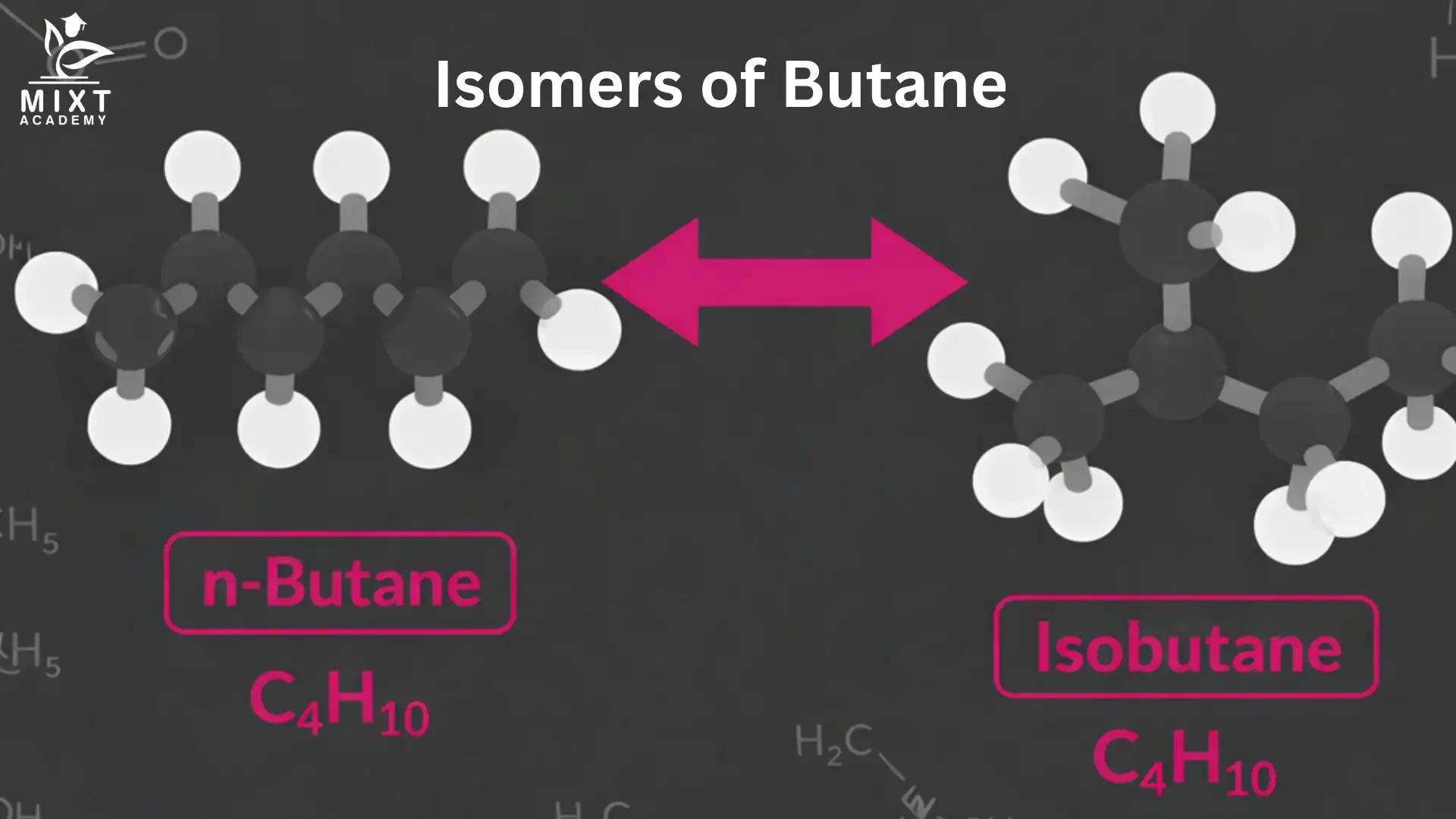
Molecular Structure of Butane (C₄H₁₀)
Butane contains four carbon atoms and ten hydrogen atoms. The carbons can be arranged in two distinct ways.
Straight-Chain Arrangement
In one form, all four carbons connect in a continuous chain.
Structural Formula: CH₃–CH₂–CH₂–CH₃
This compound is called n-butane. The “n” stands for normal or straight chain.
Branched Structure: Isobutane
Now rearrange the same four carbons.
- Three carbons form a chain
- One carbon branch from the middle carbon
Structural Formula: CH₃–CH(CH₃)–CH₃
This compound is known as isobutane, or by its systematic name, 2-methylpropane. Here lies the core n-butane and isobutane difference. The atoms stay the same. Their positions do not.
Why do These Two Isomers Behave Differently?

Structure affects physical behaviour. Key differences you should remember:
- n-Butane has a higher boiling point
- Isobutane is more compact
- Branching lowers intermolecular attraction
Measured Boiling Points:
- n-Butane: −0.5°C
- Isobutane: −11.7°C
Same formula with different behaviour.
Common Student Mistake: Confusing Rotation with Isomerism
You may see butane drawn in bent shapes. That does not mean new compounds exist. Rotation around single bonds only changes shape, not identity.
Tip For Exams:
Trace the carbon backbone with your finger.
If you never lift it, the chain is continuous.
How does this Concept Connect to Other Compounds?
Once you understand butane, larger alkanes feel easier.
- Pentane has three isomers.
- Hexane has five.
The count rises as carbon numbers increase. This idea also prepares you to understand alcohol isomerism.
Example: Isomers of Butan-1-ol differ because the OH group attaches at different carbon positions, not because the carbon count changes.
| Property | n-Butane | Isobutane |
|---|---|---|
| Structure | Straight chain | Branched |
| Carbon backbone | 4 continuous | 3 + branch |
| Boiling point | Higher | Lower |
| Shape | Less compact | Compact |
Why Teachers Emphasise This Topic?

- Visualise molecules
- Apply logic instead of memorisation
- Predict physical properties from structure
Once you master this, topics like naming rules and reaction trends become easier.
Want to learn how to train yourself for these topics?
Contact Mixt Academy for qualified online tutoring assistance according to your schedule.
Isomers of Butane – Exam-Ready Notes
Core Definition (Write This in Exams)
Butane is an alkane with the molecular formula C₄H₁₀. It shows structural isomerism because the four carbon atoms can be arranged in more than one way while keeping the same molecular formula.
Essential Concepts You Must Know
Before answering any question on butane isomers, you should recall:
- Carbon forms four covalent bonds
- Alkanes contain only single C–C bonds
- The general formula of alkanes is CnH₂n+₂
- Structural isomers differ in carbon arrangement, not formula
These points are often asked indirectly.
Structural Isomerism in Butane
Structural isomerism occurs when compounds have:
- Same molecular formula
- Different connectivity of atoms
Butane shows chain isomerism, a type of structural isomerism. This is why it falls under structural isomers of butane.
Names of Isomers of Butane
Butane has only two structural isomers:
- n-Butane
- Isobutane (2-methylpropane)
No other structural arrangement is possible for C₄H₁₀.
Labelled Structural Formula Diagrams
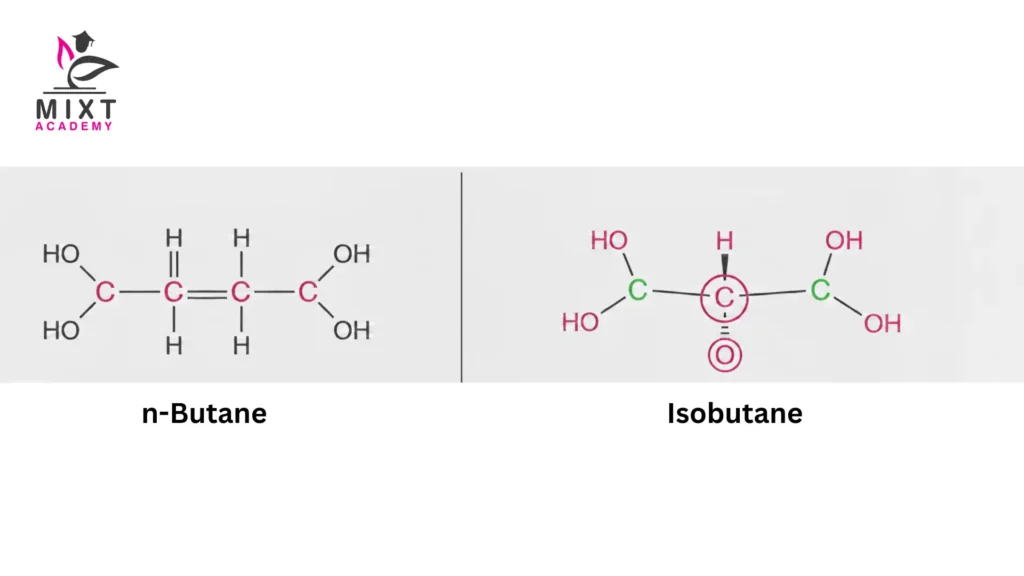
1. n-Butane (Straight-Chain Alkane)
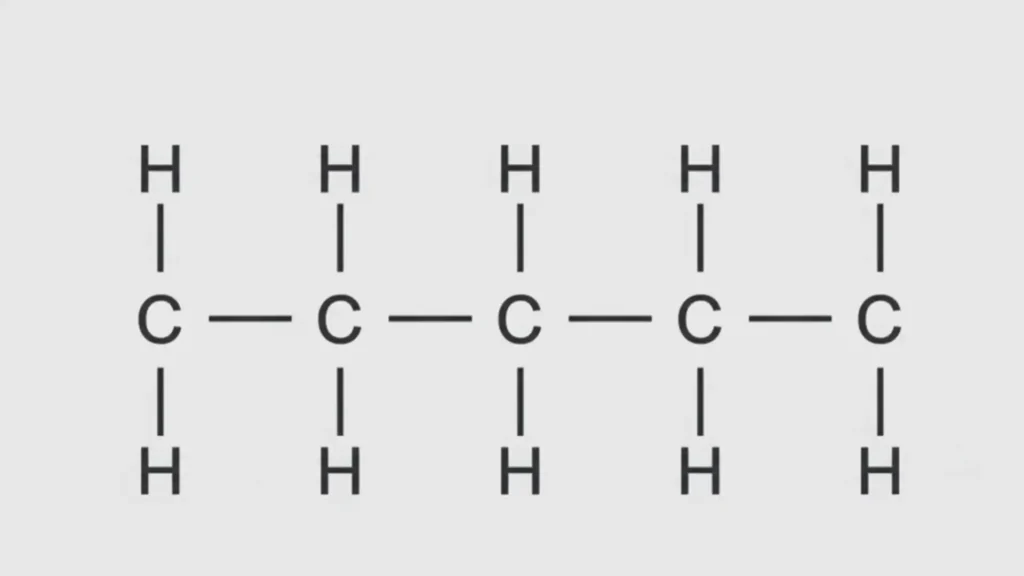
Condensed formula: CH₃–CH₂–CH₂–CH₃
Key Points:
- Four carbons in a continuous chain
- No branching
- Also called normal butane
2. Isobutane (Branched-Chain Alkane)
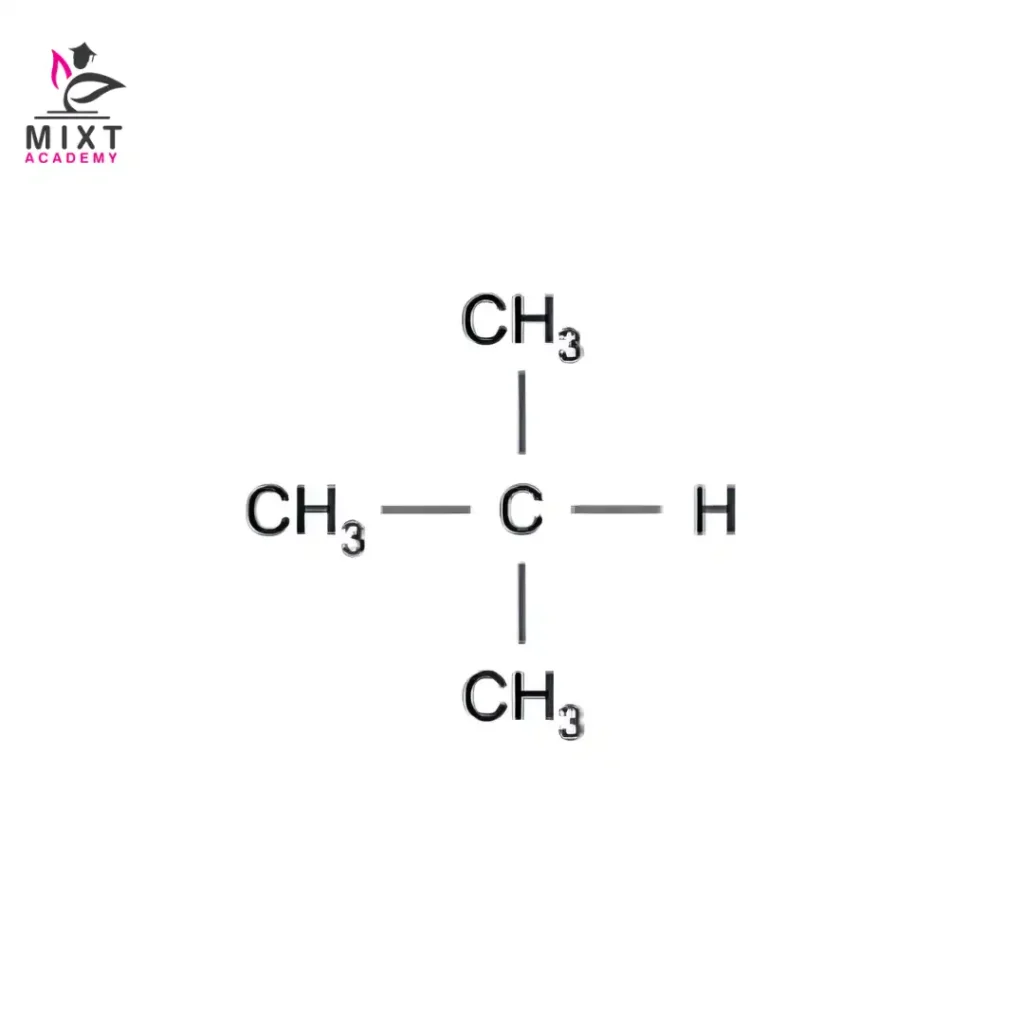
Condensed formula: CH₃–CH(CH₃)–CH₃
Systematic name: 2-methylpropane
Key Points:
- This structural change explains the n-butane and isobutane difference.
- One carbon branch from the middle carbon
- Shorter main chain
- More compact structure
Physical Property Comparison (High-Scoring Table)
Reason: Branching reduces surface contact, lowering the boiling point.
| Property | n-Butane | Isobutane |
|---|---|---|
| Structure | Straight chain | Branched |
| Type | Normal alkane | Branched alkane |
| Boiling point | −0.5°C | −11.7°C |
| Surface area | Larger | Smaller |
| Intermolecular forces | Stronger | Weaker |
Important Exam Facts (Memorise These)
- Rotation around C–C bonds does not form new isomers
- Bent drawings of butane still represent the same molecule
- Only changes in the carbon backbone create new isomers
- Butane has the minimum carbon number where chain isomerism appears
Link to Other Compounds (Concept Extension)
Understanding butane helps you later with:
- Pentane and hexane isomer counting
- Alcohol isomerism, such as isomers of Butan-1-ol, where the OH group position changes
- IUPAC naming rules are based on the longest carbon chain
Learn Isomers of Butane the Smart Way with Mixt Academy.
Struggling to visualise structural isomers of butane or understand the n-butane and isobutane? Mixt Academy is an online tutoring platform that simplifies organic chemistry through concept-based teaching, exam-focused notes, and one-on-one guidance. Learn at your pace, clear doubts instantly, and build strong fundamentals that boost exam confidence.
Finding Chemistry Concepts Hard to Learn?
Expert chemistry concepts like isomers of butane with Mixt Academy’s qualified tutors.
Why Are the Isomers of Butane Important to Understand?
Understanding the isomers of butane helps you see why structure matters more than memorising formulas. Although n-butane and isobutane share the same molecular formula, their different carbon arrangements lead to clear differences in physical properties like boiling point.
This idea explains the core of structural isomers of butane and prepares you for harder topics such as isomer counting, IUPAC naming, and even isomers of Butan-1-ol. Once you learn the difference between n-butane and isobutane, organic chemistry becomes manageable rather than confusing. In exams, this topic tests understanding, not memory, making it a high-scoring and concept-building area.