This resource, created for IGCSE and Class 10 students, uses simple but thorough concepts illustrated with very clear explanations and real-life examples. Whether you are revising MCQs, working on practice papers, or preparing for past papers focused on acids, bases, and salts, this comprehensive blog by Mixt Academy is sure to improve your understanding as well as your self-confidence.
What are Acids, Bases, and Salts?
First of all, acids, bases, and salts are the main classes of compounds in chemistry, considered to be little more than their specific properties as defined by the way they react with each other. Here, you will find a more detailed classification of each of these compounds.
What are Acids?

Acids are a category of chemical compounds that, when dissolved in water, produce hydrogen ions (H⁺) as the primary species. They usually have a sour taste, might corrode or be sticky, and change the colour of blue litmus paper to red. Acids are characterised by their low pH (less than 7), and such acids are called acid-base reactions.
The most common examples of acids are ascorbic acid (present in oranges) and hydrochloric acid (found in stomach acid). Acids may be strong (like hydrochloric acid) or weak (like lactic acid in milk), depending on the acid’s character.
Key Characteristics of Acids
- Taste: Sour (found in fruits like lemons and sour candies).
- Touch: Can feel corrosive or sticky, depending on the strength.
- pH: Below 7 (with 7 being neutral; bases have a pH above 7).
- Litmus Test: Turns blue litmus paper red.
- Reactivity: Acids react with metals (e.g., zinc reacting with hydrochloric acid to produce hydrogen gas) and with bases to undergo neutralisation reactions.
Theories of Acidity
- Arrhenius Theory: Acids are substances that increase the concentration of H⁺ ions (protons) in water, forming hydronium ions (H₃O⁺).
- Bronsted-Lowry Theory: Acids are proton donors; they donate H⁺ ions in chemical reactions.
- Lewis Theory: Acids are electron pair acceptors, a broader definition that includes substances that can accept pairs of electrons from other substances.
Examples & Types of Acids
- Strong Acids: These acids completely dissociate in water to produce H⁺ ions.
Examples include hydrochloric acid (HCl) and sulphuric acid (H₂SO₄). - Weak Acids: These acids dissociate only to a limited extent in water, which indicates that they do not release all of their H⁺ ions.
Examples are acetic acid (found in vinegar) and carbonic acid (found in carbonated drinks). - Mineral Acids: These are inorganic acids commonly found in industry and biology.
Examples include sulphuric acid (H₂SO₄), nitric acid (HNO₃), and hydrochloric acid (HCl). - Organic Acids: These acids contain carbon and often come from natural sources. Carboxylic acids, such as citric acid (found in citrus fruits), are common examples.
Struggling to Differentiate Between Acids or Remember Their Properties?
Mixt Academy tutors break these concepts into simple, exam-friendly steps—so you learn faster and remember longer.
What Are Bases?

A base is a substance that releases hydroxide ions (OH⁻) when dissolved in water or accepts hydrogen ions (H⁺) during a chemical reaction. Bases typically have a bitter taste, feel slippery or soapy to the touch, and have a pH value greater than 7.
They turn red litmus paper blue and react with acids in neutralisation reactions to form salt and water. Common examples of bases include sodium hydroxide (NaOH) and ammonia (NH₃). Bases are classified as strong or weak depending on how completely they dissociate in water.
Key Characteristics of Bases
- Taste: Bitter (not edible; do not taste in experiments)
- Touch: Slippery or soapy feel
- pH Level: Above 7 (7 is neutral; acids are below 7)
- Litmus Test: Turns red litmus paper blue
- Reactivity: Reacts with acids to form salt and water (neutralisation)
Theories of Bases
- Arrhenius Theory: Bases are substances that produce hydroxide ions (OH⁻) in aqueous solutions.
- Bronsted–Lowry Theory: Bases are proton acceptors; they accept hydrogen ions (H⁺) during chemical reactions.
- Lewis Theory: Bases are electron pair donors, capable of donating a pair of electrons to form a chemical bond.
Examples and Types of Bases
- Strong Bases: Completely dissociate in water to produce ions.
Examples: Sodium hydroxide (NaOH), Potassium hydroxide (KOH) - Weak Bases: Partially dissociate in water.
Examples: Ammonia (NH₃), Magnesium hydroxide - Alkalis: Bases that are soluble in water.
Examples: Sodium hydroxide (NaOH), Potassium hydroxide (KOH), Calcium hydroxide - Insoluble Bases: Bases that do not dissolve in water.
Examples: Copper(II) oxide (CuO), Iron(III) oxide (Fe₂O₃)
Everyday Uses of Bases
- Soap and Detergents: Used for cleaning due to their slippery nature.
- Toothpaste: Neutralises acids in the mouth.
- Baking Soda (Sodium Bicarbonate): Used in baking and as an antacid.
- Drain Cleaners and Bleach: Strong bases are used for heavy cleaning.
What Are Salts?

Salts are a broad group of ionic substances produced when an acid reacts with a base in a chemical neutralisation process. They are made up of positively charged ions (cations) and negatively charged ions (anions) that attract each other through strong ionic forces, forming a compound with no overall charge.
Although many people think only of table salt, salts include a wide variety of substances used in food, medicine, agriculture, and industry. Most salts exist as crystalline solids with distinct physical and chemical properties.
Key Characteristics of Salts
- Ionic Makeup: Composed of cations (often metals like Ca²⁺ or NH₄⁺) and anions (such as NO₃⁻ or CO₃²⁻).
- Method of Formation: Commonly produced through acid–base neutralisation
(Acid + Base → Salt + Water) - Physical Structure: Usually form hard, crystalline solids due to strong attractions between oppositely charged ions.
- Electrical Neutrality: The total positive and negative charges balance, so the compound has no net charge.
Examples of Salts
- Calcium Carbonate (CaCO₃): Found in limestone and chalk; used in construction and as an antacid.
- Ammonium Nitrate (NH₄NO₃): Used in fertilisers and explosives.
- Copper(II) Sulfate (CuSO₄): A blue crystalline salt used in laboratories and agriculture.
- Sodium Fluoride (NaF): Added to toothpaste and drinking water to protect teeth.
Properties of Salts
- Electrical Conductivity: Conducts electricity when dissolved in water or melted, as ions are free to move.
- High Melting Points: Require large amounts of energy to break the strong ionic bonds.
- Variable Solubility: Some dissolve easily in water, while others are nearly insoluble.
Reactions Involving Acids, Bases, and Salts
Acids, bases, and salts interact in various reactions, with neutralisation being the most common. In a neutralisation reaction, an acid and a base react to form a salt and water. For example, HCl (hydrochloric acid) reacts with NaOH (sodium hydroxide) to produce NaCl (sodium chloride) and H₂O (water). Acids also react with metal carbonates to produce a salt, water, and carbon dioxide.
An example is the reaction between CaCO₃ (calcium carbonate) and HCl, forming CaCl₂ (calcium chloride), H₂O, and CO₂. Salts can further react with stronger acids or bases, or with each other in a double displacement reaction, often forming new salts or precipitates, depending on the solubility and strength of the reactants.
Key Reactions that Involve Acids, Bases and Salts
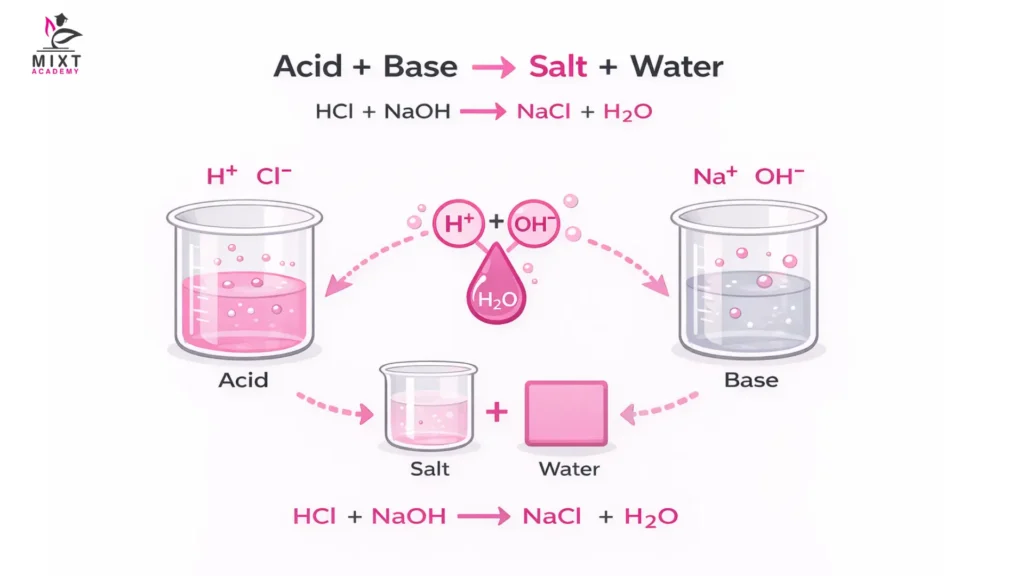
Acid + Base (Neutralisation)
Definition: In this reaction, an acid donates protons (H⁺), and a base accepts them, resulting in the formation of a salt and water.
Example: Hydrochloric acid (HCl) + Sodium hydroxide (NaOH) → Sodium chloride (NaCl) + Water (H₂O)
General Form: Acid + Base → Salt + Water
Acid + Metal Carbonate/Bicarbonate
Definition: When an acid reacts with a metal carbonate or bicarbonate, the products are a salt, water, and carbon dioxide gas.
Example: Calcium carbonate (CaCO₃) + Hydrochloric acid (HCl) → Calcium chloride (CaCl₂) + Water (H₂O) + Carbon dioxide (CO₂)
Acid + Metal Oxide
Definition: A metal oxide (which acts as a base) reacts with an acid to produce a salt and water.
Example: Copper (Cu) oxide (O) + Sulfuric acid (H₂SO₄) → Copper sulfate (CuSO₄) + Water (H₂O)
Salt Reactions
With Acids/Bases
A salt can react with a stronger acid or base, especially if it was formed from a weaker acid or base. The reaction can produce a different salt or regenerate the original acid or base.
Double Displacement
Salts can also react with other salts, particularly in solution, to form new salts. A common example of this occurs when a precipitate (an insoluble solid) is formed.
How Are Salts Formed and Characterised?
Salts are characterised by the combination of the acid and base used in their formation. Here are some examples of how salts are formed:
- Strong Acid + Strong Base → Neutral Salt (e.g., NaCl, sodium chloride).
- Strong Acid + Weak Base → Acidic Salt (e.g., NH₄Cl, ammonium chloride).
- Weak Acid + Strong Base → Basic Salt (e.g., CH₃COONa, sodium acetate).
“Why Do Students Struggle to Remember Salt Types in Exams?”
Mixt Academy certified chemistry tutors simplify salt formation using easy rules and guided practice, helping students identify salt types quickly and confidently.
Indicators and Salt Solutions
Unlike acids and bases, which affect litmus paper (acids turn blue litmus paper red, and bases turn red litmus paper blue), salt solutions generally do not cause a colour change in litmus paper. This is because salts are typically neutral and do not significantly alter the hydrogen ion concentration.
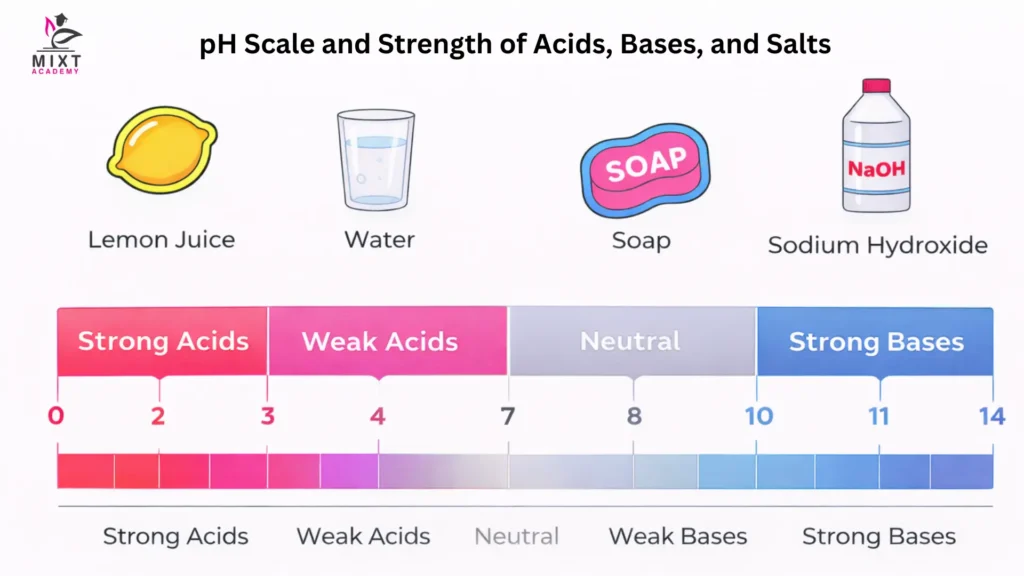
The pH Scale and Strength of Acids and Bases
The pH scale is used to measure the acidity or basicity of a substance. It ranges from 0 to 14:
- pH 7 is neutral (pure water).
- pH below 7 indicates an acid.
- pH above 7 indicates a base (alkali).
The lower the pH, the stronger the acid. For example:
- Hydrochloric acid (pH ≈ 1) is very strong.
- Lemon juice (pH ≈ 2–3) is weaker.
Similarly, the higher the pH, the stronger the base. For example:
- Sodium hydroxide (pH ≈ 13–14) is a strong base.
- Soap solution (pH ≈ 9–10) is a weaker base.
Each change of one unit on the pH scale represents a tenfold change in acidity or alkalinity, making it a logarithmic scale.
The pH Scale and Strength of Acids, Bases, and Salts
| Substance | Key Properties | Example |
|---|---|---|
| Acids | Sour taste, pH < 7, turns blue litmus red, releases H⁺ ions | Lemon juice, HCl |
| Bases | Bitter, slippery, pH > 7, turns red litmus blue, releases OH⁻ ions | NaOH, soap |
| Salts | Usually neutral, crystalline solids, formed by neutralisation | NaCl, CuSO₄ |
What are Indicators and Synthetic Indicators?
Indicators are substances that help determine whether a solution is acidic, basic, or neutral by changing colour. These tools are vital in chemistry, allowing for the identification of a substance’s pH. There are two primary types of indicators: natural and synthetic.
What are Indicators?
Indicators are chemical substances that change their colour based on the pH level of a solution. This colour shift occurs as the indicator reacts with hydrogen ions (H⁺) in acidic solutions or hydroxide ions (OH⁻) in basic solutions, providing a visual representation of whether the solution is acidic, neutral, or alkaline.
What are Synthetic Indicators?
Synthetic indicators are artificial substances, usually made from complex organic compounds, that change colour in response to specific chemical conditions, typically at certain pH levels. These are commonly used in laboratory experiments and titrations for accurate pH measurement.
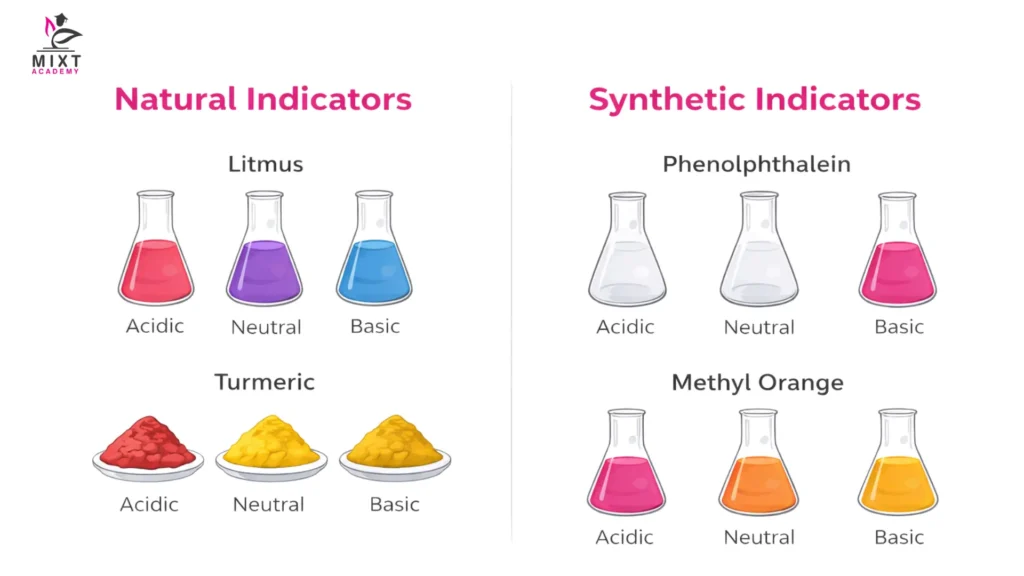
Natural vs Synthetic Indicators
| Type of Indicator | Definition | Characteristics | Examples |
|---|---|---|---|
| Natural Indicators | Sourced from nature, such as plants and lichens | Often less precise with gradual colour changes; commonly used in simple tests | Litmus: Red in acid, blue in base Turmeric: Red in alkaline solutions |
| Synthetic Indicators | Man-made substances created in laboratories | Provide sharp, distinct colour changes; ideal for precise chemical reactions | Phenolphthalein: Colourless in acid, pink in base Methyl Orange: Red in acid, yellow in base, orange in neutral |
Exam Preparation: MCQs and Practice Questions
This section is designed to help IGCSE and Class 10 students test their understanding of Acids, Bases, and Salts and prepare effectively for exams. Practising MCQs and structured questions improves accuracy, speed, and confidence for both school tests and board exams.
Multiple Choice Questions (MCQs)
1. Which of the following has a pH value of 2?
- A. Soap solution
- B. Pure water
- C. Lemon juice
- D. Sodium hydroxide
Answers: C
2. A substance that turns red litmus paper blue is most likely a:
- A. Salt
- B. Acid
- C. Base
- D. Neutral solution
Answers: 2–C
3. Which pair will undergo a neutralisation reaction?
- A. HCl and NaCl
- B. NaOH and HCl
- C. CuSO₄ and H₂O
- D. CaCO₃ and NaCl
Answers: 3–B
4. When an acid reacts with a metal carbonate, the products are:
- A. Salt and water
- B. Salt and hydrogen
- C. Salt, water, and carbon dioxide
- D. Water and carbon dioxide
Answers: 4–C
5. Which of the following is a weak acid?
- A. Hydrochloric acid
- B. Sulfuric acid
- C. Nitric acid
- D. Acetic acid
Answers: 5–D
Few Important Practice Questions (Short & Long Answer)
- Define an acid and a base using the Arrhenius theory.
- Write the balanced chemical equation for the reaction between:
a) Hydrochloric acid and sodium hydroxide
b) Sulfuric acid and copper oxide - Explain why sodium chloride is considered a neutral salt.
- State two differences between strong acids and weak acids.
- Describe the pH scale and explain why it is called a logarithmic scale.
- Name one natural indicator and one synthetic indicator. State their colour changes in acidic and basic solutions.
- A student adds dilute hydrochloric acid to calcium carbonate.
a) Name the gas produced.
b) Write the word equation for the reaction.
c) State one test to identify the gas.
Why Are Acids, Bases, and Salts Important to Understand?
Understanding Acids, Bases and Salts is important for mastering core chemistry concepts in and Class 10. These substances explain everyday phenomena, from digestion and cleaning to food preservation and medicine. By learning their properties, reactions, and formation, along with the pH scale and indicators, students can confidently identify acidic, basic, and neutral substances.
Neutralisation reactions show how acids and bases interact to form useful salts, linking theory to real-life applications. With expert guidance and structured practice on the Mixt Academy online tutoring platform, students can improve these concepts through MCQs and exam-style questions. Mastering this topic not only improves academic performance but also deepens understanding of the chemical processes shaping the world around you.