A physical measurement consists of two parts: a numeric value that shows the quantity and a measurement unit that defines the measurement standard. The physical quantity is defined by these components, which give the expression Q = n × u.
For instance, the length of an object can be expressed as 40″ cm” because 40 serves as the numerical value, while cm (centimetres) functions as the unit. The product of a physical quantity’s value and its unit system remains unchanged when the quantity is measured using two distinct unit systems. The equivalences 45″ min” = 2700″ s” and 3.5 L = 3500 mL serve as examples.
Mixt Academy is here to help you achieve your academic goals.
A unit is a standard measure of a physical quantity. It tells us in what terms the measurement is expressed.
Unit Examples
The unit of length is the meter, which has the abbreviation (m). The unit of mass is the kilogram, which has the abbreviation (kg). The unit of time is the second, which has the abbreviation (s).
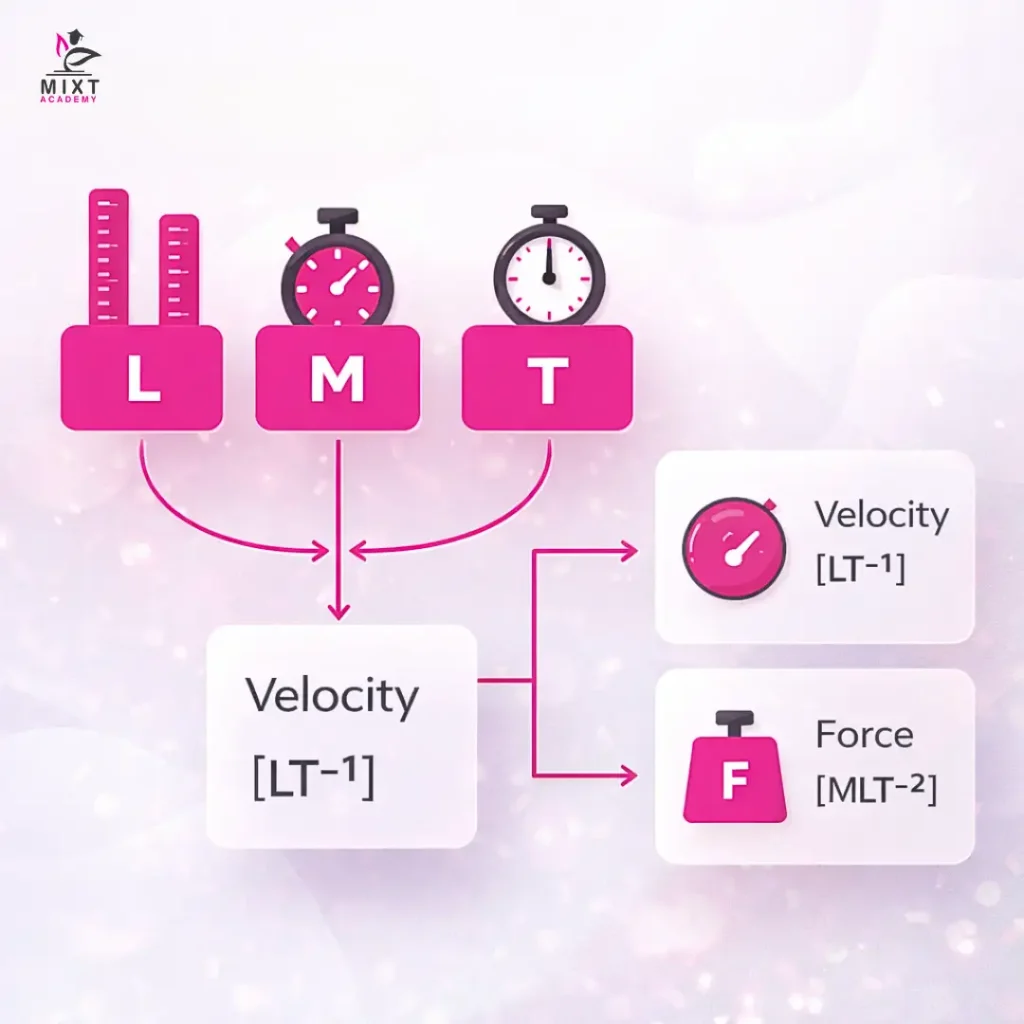
Dimensions define the characteristics of a physical quantity using basic units, such as length (L), mass (M), and time (T). The dimension of velocity shows its representation through [LT^(-1)] while the dimension of force shows its representation through [MLT^(-2) ].
Dimensional analysis involves studying how physical quantities relate to one another by identifying their dimensions.
Dimensional Formula
The dimensional formula shows how a derived quantity relates to fundamental units through the specific powers of those units. The dimensional formula for the derived quantity Q is Q = M^a L^b T^c; the term M^a L^b T^c is its dimensional formula, where the exponents a, b, and c are called the dimensions of the quantity.
Don’t let small gaps turn into big problems in exams. Get guided learning with real tutors who simplify every concept.
Dimensional Constants
Physical quantities that have fixed values and possess dimensions are called dimensional constants. Examples include the gravitational constant G, Planck’s constant h, the universal gas constant R, and the speed of light in a vacuum c.
Dimensionless Quantities

Dimensionless quantities refer to physical, chemical, or mathematical values that lack measurement units and possess a net dimension of one.
Dimensional Variables
Quantities with dimensions whose values can vary are called dimensional variables. Common examples include velocity, acceleration, force, work, and power.
Dimensionless Variables
Dimensionless variables are physical quantities that have no dimensions and whose values can vary. Examples include specific gravity, refractive index, coefficient of friction, and Poisson’s ratio.
Stop memorising, start understanding and get quality revision material.

For any physically meaningful equation relating physical quantities, the dimensions of every term on both sides must be identical.
Consider a physical quantity with dimensions a, b, and c corresponding to length (L), mass (M), and time (T), respectively. If n_1is the numerical value of Qin a system with fundamental units L_1, M_1, and T_1, and n_2 is its numerical value in another system with fundamental units L_2, M_2, and T_2, then the values are related by:

We teach smart shortcuts and patterns so you don’t have to memorise everything blindly.
| Physical Quantity | SI Unit | Symbol |
|---|---|---|
| Frequency | hertz | Hz |
| Energy | joule | J |
| Force | newton | N |
| Power | watt | W |
| Pressure | pascal | Pa |
| Electric charge (quantity of electricity) | coulomb | C |
| Electric potential difference / EMF | volt | V |
| Electric resistance | ohm | Ω |
| Electric conductance | siemens | S |
| Electric capacitance | farad | F |
| Magnetic flux | weber | Wb |
| Inductance | henry | H |
| Magnetic flux density | tesla | T |
| Illumination | lux | lx |
| Luminous flux | lumen | lm |

| Physical Quantity | Unit | Dimensional Formula |
|---|---|---|
| Acceleration (or gravity) | m/s² | LT⁻² |
| Angle / Angular displacement | rad | MoLoTo |
| Angular frequency | rad/s | T⁻¹ |
| Angular impulse (torque × time) | N·m·s | ML²T⁻¹ |
| Angular momentum (Iω) | kg·m²/s | ML²T⁻¹ |
| Angular velocity | rad/s | T⁻¹ |
| Area | m² | L² |
| Boltzmann constant | J/K | ML²T⁻²θ⁻¹ |
| Bulk modulus | Pa | ML⁻¹T⁻² |
| Calorific value | J/kg | L²T⁻² |
| Coefficient of linear/areal/volume expansion | °C⁻¹ or K⁻¹ | θ⁻¹ |
| Surface tension (force/length) | N/m | MT⁻² |
| Thermal conductivity | W/m·K | MLT⁻³θ⁻¹ |
| Viscosity | poise | ML⁻¹T⁻¹ |
| Compressibility (1/bulk modulus) | Pa⁻¹ | M⁻¹LT² |
| Density | kg/m³ | ML⁻³ |
| Displacement / Wavelength / Focal length | m | L |
| Electric capacitance (charge/potential) | F | M⁻¹L⁻²T⁴I² |
| Electric conductance | S | M⁻¹L⁻²T³I² |
| Electric conductivity | S/m | M⁻¹L⁻³T³I² |
| Electric charge | C | IT |
| Electric current | A | I |
| Electric dipole moment | C·m | LTI |
| Electric field strength (force/charge) | N/C | MLT⁻³I⁻¹ |
| Electric resistance | Ω | ML²T⁻³I⁻² |
| EMF / Electric potential | V | ML²T⁻³I⁻¹ |
| Energy | J | ML²T⁻² |
| Energy density | J/m³ | ML⁻¹T⁻² |
| Entropy | J/θ | ML²T⁻²θ⁻¹ |
| Force | N | MLT⁻² |
| Force constant / Spring constant | N/m | MT⁻² |
| Frequency | Hz | T⁻¹ |
| Gravitational potential | J/kg | L²T⁻² |
| Heat / Energy | J | ML²T⁻² |
| Illumination / Illuminance | lux | MT⁻³ |
| Impulse | N·s | MLT⁻¹ |
| Inductance | H | ML²T⁻²I⁻² |
| Intensity of gravitational field | N/kg | LT⁻² |
| Intensity of magnetisation | A/m | L⁻¹I |
| Joule’s constant | J/cal | MoLoTo |
| Latent heat | J/kg | L²T⁻² |
| Linear density | kg/m | ML⁻¹ |
| Luminous flux | lumen | ML²T⁻³ |
| Magnetic dipole moment | A·m² | L²I |
| Magnetic flux | Wb | ML²T⁻²I⁻¹ |
| Magnetic induction | T | MT⁻²I⁻¹ |
| Magnetic pole strength | A·m | LI |
| Modulus of elasticity | Pa | ML⁻¹T⁻² |
| Moment of inertia | kg·m² | ML² |
| Momentum | kg·m/s | MLT⁻¹ |
| Permeability of free space | H/m | MLT⁻²I⁻² |
| Permittivity of free space | F/m | M⁻¹L⁻³T⁴I² |
| Planck’s constant | J·s | ML²T⁻¹ |
| Poisson’s ratio | – | MoLoTo |
| Power | W | ML²T⁻³ |
| Pressure | Pa | ML⁻¹T⁻² |
| Pressure coefficient / Volume coefficient | °C⁻¹ | θ⁻¹ |
| Pressure head | m | MoLTo |
| Radioactivity | disintegrations/s | MoLoT⁻¹ |
| Ratio of specific heats | – | MoLoTo |
| Refractive index | – | MoLoTo |
| Resistivity | Ω·m | ML³T⁻³I⁻² |
| Specific conductance | S/m | M⁻¹L⁻³T³I² |
| Specific entropy | 1/K | M⁻¹L⁻²T²θ |
| Specific gravity | – | MoLoTo |
| Specific heat | J/kg·θ | L²T⁻²θ⁻¹ |
| Specific volume | m³/kg | M⁻¹L³ |
| Speed | m/s | LT⁻¹ |
| Stefan’s constant | W/m²·θ⁴ | MLT⁻³θ⁻⁴ |
| Strain | – | MoLoTo |
| Stress | Pa | ML⁻¹T⁻² |
| Surface energy density | J/m² | MT⁻² |
| Temperature | °C or θ | MoLoToθ |
| Temperature gradient | °C/m | MoL⁻¹Toθ |
| Thermal capacity | J/θ | ML²T⁻²θ⁻¹ |
| Time period | s | T |
| Torque / Moment of force | N·m | ML²T⁻² |
| Universal gas constant | J/mol·θ | ML²T⁻²θ⁻¹ |
| Universal gravitational constant | N·m²/kg² | M⁻¹L³T⁻² |
| Velocity | m/s | LT⁻¹ |
| Velocity gradient | s⁻¹ | T⁻¹ |
| Volume | m³ | L³ |
| Water equivalent | kg | MoLoTo |
| Work | J | ML²T⁻² |
Let Mixt Academy guide you every step of the way.
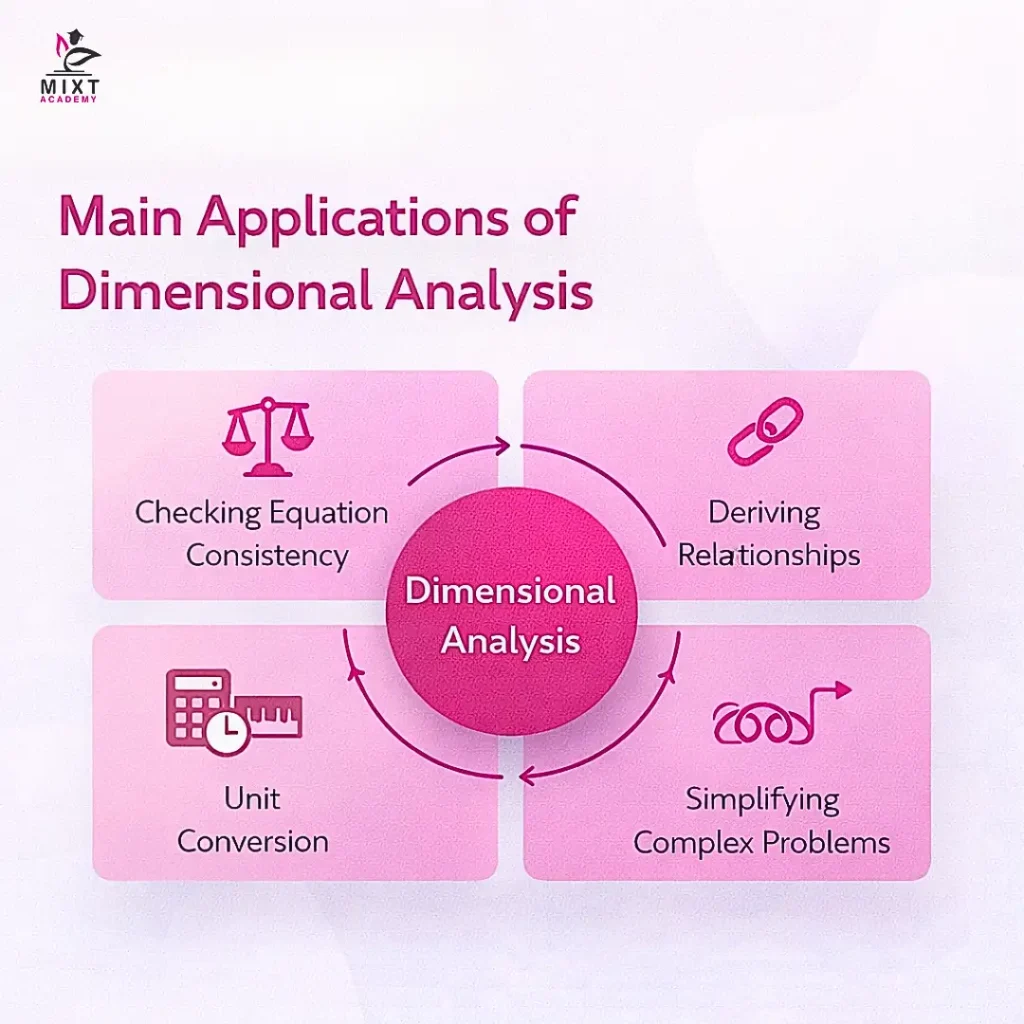
Dimensional analysis is a powerful tool in physics for understanding and working with physical quantities. It helps simplify complex problems and ensure consistency in equations. Fourier established the foundations of dimensional analysis, and it has several important applications:
Unit Conversion:
Dimensional analysis provides a systematic way to convert a physical quantity from one system of units to another.

Want to score better in Physics exams? Mixt Academy offers expert-led learning on Units and Dimensions, helping you master dimensional formulas, constants, and unit conversions. Our qualified tutors focus on exam-relevant concepts and problem-solving techniques that save time and reduce errors. Strengthen your Physics fundamentals with Mixt Academy. Join today and book a demo session to get started.
In physics, a dimension describes the nature of a physical quantity in terms of fundamental units such as mass (M), length (L), and time (T). It shows how a derived quantity relates to these basics, but it does not indicate the magnitude of the quantity.
If you want to learn more and strengthen your physics concepts, join Mixt Academy, a trusted online tutoring platform.
Force is defined as F = ma, where m is mass and a is acceleration. Mass is a fundamental quantity [M], and acceleration has the dimension [LT^(-2) ]. Therefore, the dimension of force is:
[F]=[M][LT^(-2)]=[MLT^(-2) ]
Dimensional analysis is a method for checking equations or relationships among physical quantities. It is based on the principle that only quantities with the same dimensions can be compared, added, or equated. For example, kinetic energy can be compared with potential energy, but not with force or acceleration.
Write all quantities in terms of M, L, T. Express the RHS with unknown powers, equate the dimensions of both sides, and solve the resulting system of equations to find the exponents.

Mixt Academy is a global online tutoring platform that connects students with expert IGCSE, GCSE, and A-Level tutors for one-to-one learning. With flexible scheduling, personalized lesson plans, and experienced teachers from top curricula, Mixt Academy helps students strengthen concepts, improve exam skills, and achieve higher grades with confidence.
Understanding Units and Dimensions of Physical Quantities A physical measurement…
Ultimate Guide to Vocabulary Building Techniques for Effective Language Learning…
IGCSE Physics Formula Sheet: Key Formulas for 0625 & Edexcel…
IGCSE Chemistry Papers: Common Mistakes & Exam Tips This IGCSE…
IGCSE Extended vs Core Tiers: How to Pick the Right…
GCSE vs A-Levels Student Friendly Guide Selecting educational credentials is…
Understanding GCSE Grade Boundaries and How to Prepare? Grade boundaries…
Differences GCSE English Language vs English Literature GCSE English is…
A Level vs AP: Complete Guide to Choosing the Right…
Top Benefits of Completing AS and A Levels Privately Explore…
Cambridge vs IB Guide to Choosing the Best Curriculum Choosing…
IB vs A Levels: Which Qualification Is Right for You?…
How to Use OCR Past Papers for Effective Revision? Past…
AQA Physics Equation Sheet: Complete GCSE Guide for Combined Science…
AQA vs Edexcel Complete GCSE Comparison and Guide Choosing between…
Complete Guide to the AQA GCSE Chemistry Specification Understanding the…
Hire an Expert Tutor from Just 15$/hr